All formal charges must be displayed on the molecule: The correct structure for the `NO_3^-` molecule is thereforeĭetermine the formal charge of each of the atoms. We have to consider these formal charges in the structure of this molecule. Remember that the total formal charge must be equal to the charge on the molecule. This corresponds to the negative charge in the molecule itself. In total, the molecule has a formal charge of -1: +1 from the `N` and 2(-1) from the two single bonded O. The formal charge of a double bonded oxygen atom is lower than that of a single bonded oxygen atom, indicating stability. This is why you'll find `O` double bonded most of the time, as opposed to single bonded. Now let's do the formal charge calculations for the 2 `O` atoms with single bonds:Īnd finally, the formal charge of the `O` with a double bond: That is what the formal charge indicates. Even though `N` now has a filled valence shell, it had to share 1 electron in order to get there. Since `N` by itself has 5 valence electrons, it has effectively lost one electron, leaving it with a +1 formal charge. You can think of it this way: nitrogen has access to 8 electrons but in reality only "owns" 4 of those electrons. In other words, even though `N` has a fulfilled octet, its overall "charge" is +1. This means that the formal charge of `N` is +1. The formal charge for `N` is calculated as such: Nonetheless, just know that each straight line is a bond and each dot is an electron.įirst, notice that the octets are satisfied for each of the atoms. We haven't formally discussed Lewis structures yet (they're later on in this post), so it may help to come back to this section while you're going through the next post. For a charged molecule such as `NH_4^+`, the total formal charge must be equal to the charge of +1`įor example, let's calculate the formal charges of the atoms in this atom. For example, `CH_4` must have a total formal charge of 0, since there's no charge on the `CH_4` molecule. The total formal charge of the constituent atoms must be equal to the overall charge of the molecule. A formal charge that's too high or too low is unlikely to exist.Ģ. Atoms like to minimize formal charge if possible. There are two general rules for formal charges:ġ. This is because formal charge looks at the charge of the individual atom and whether or not that atom has effectively gained or lost electrons. In calculating formal charge, each bond counts as one electron. In other words, an atom with satisfied valency may have access to 8 electrons, but may want to own more electrons for itself.Įlectrons in the formal charge calculation are counted differently than electrons are in valency. By taking away this concept and idealizing bonds as equal sharings of electrons, we can determine the number of electrons that an atom "owns." Even though an atom has satisfied valency, it can still have a formal charge because formal charge indicates how many electrons are belonging to that atom. We learned in the previous sections that bonds usually contain a dipole moment where electrons in the bond are oriented closer to the more electronegative atom. The significant of the formal charge is that it takes away the emphasis of bond polarity. 
We call this the formal charge of the atom, which is represented by a number above the atom. The assumption behind formal charge is that atoms, in the process of satisfying valency, may end up with more electrons than they would have by themselves. Atoms will try to minimize formal charge (see below section)īefore we begin with Lewis structures, we have to discuss formal charge. 
The most electronegative atom will be near the center of the molecule.Ĥ. Only valence electrons are involved in bonding.ģ. Each bond consists of 2 shared valence electrons.Ģ. There are a couple assumptions we make in Lewis structures:ġ. 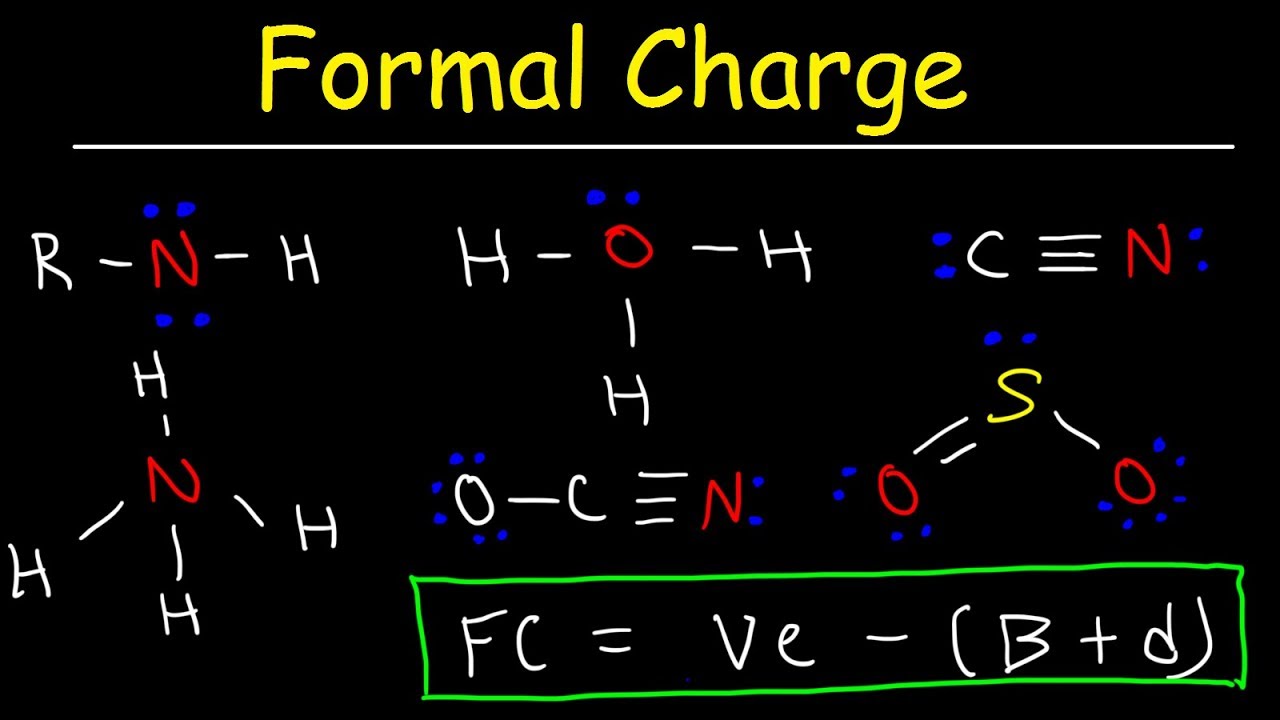
Lewis structures revolve around electrons and satisfying valency. Lewis dot structures allow us to visualize the general bonding and 2d orientation of a molecule. Up until now we've only looked at bonding between atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
